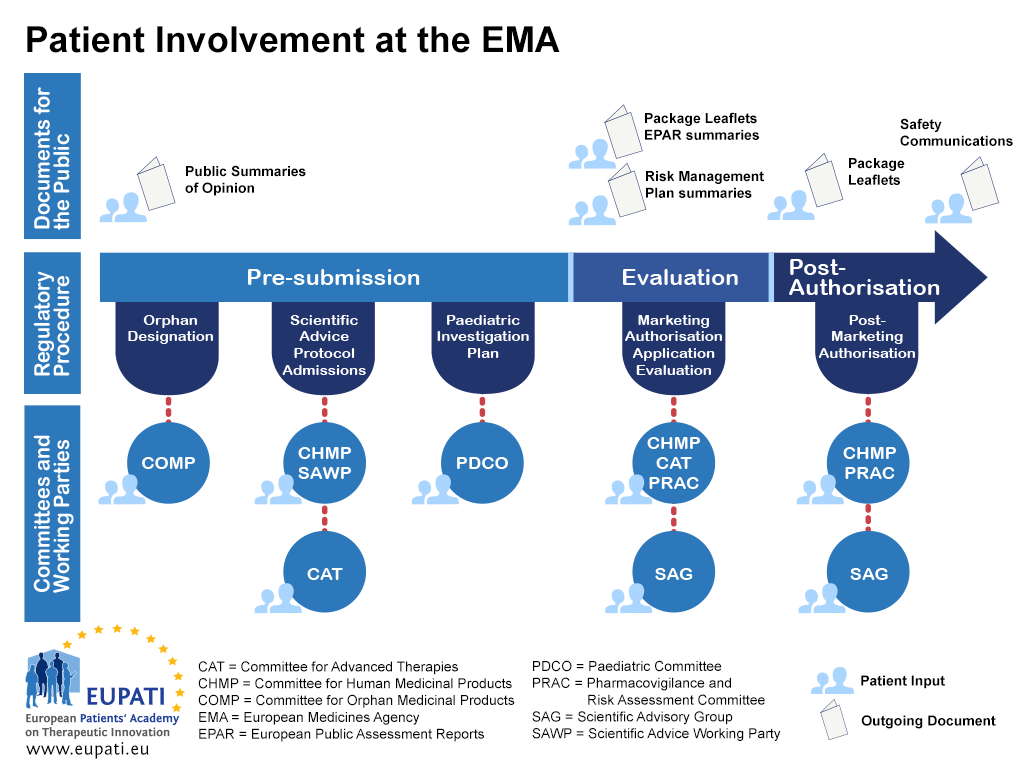
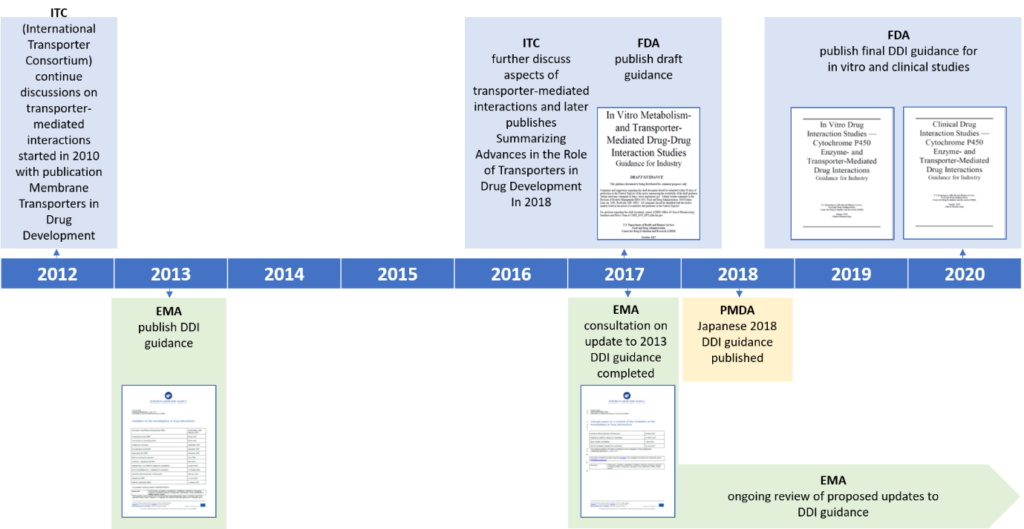
Frontiers | A Regulatory Risk-Based Approach to ATMP/CGT Development: Integrating Scientific Challenges With Current Regulatory Expectations

EMA publishes safety monitoring plan and guidance on risk management planning for COVID-19 vaccines | European Medicines Agency
European Medicines Agency pos-authorisation procedural advice for the users of the centralised procedure

Guidance to sponsors on how to manage clinical trials during the COVID-19 pandemic | European Medicines Agency

SIFO - Società Italiana di Farmacia Ospedaliera e dei servizi farmaceutici delle aziende sanitarie - Prevenire la carenza di farmaci: la guida dell'EMA alle buone pratiche

GUIDANCE ON THE MANAGEMENT OF CLINICAL TRIALS DURING THE COVID-19 (CORONAVIRUS) PANDEMIC vs 4 : KEY CHANGES - Formiventos